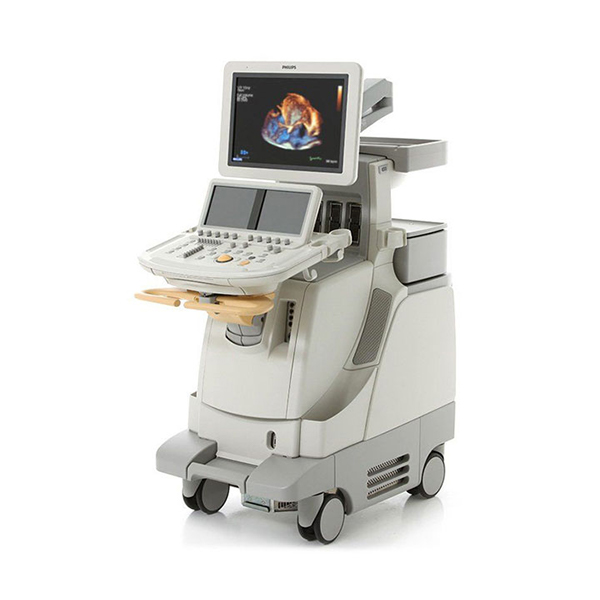
Philips iE33 Ultrasound Machine –
MPIN: MP11726
Sign in to view priceAsk for Quote
Overview
Application training for the Philips iE33
KPI’s on-staff sonographer can provide onsite applications training or remote training via videoconference at a set price plus travel costs. A pre-recorded video training course is included in the sale, lease or rental of the Philips iE33 from KPI Healthcare.
Philips iE33 Service options
Free technical support is available from KPI during installation and over the course of the standard limited warranty. Technical support is available after the warranty period at an hourly cost per issue.
Philips iE33 Maintenance
KPI recommends the use of a surge protector along with a dedicated power outlet. Probes should be disinfected after every use with a disinfectant wipe proven not to damage the lens (KPI recommends SonoWipes for this.) KPI recommends one PM visit (preventative maintenance) every year.
Philips iE33 Dimensions & Weight
Height: (adjustable, maximum) 1626 mm (64 in), (minimum) 1397 mm (55 in)
Width: 559 mm (22.0 in)
Depth: 1092 mm (43 in)
Weight: (no Peripherals) 150kg (331lbs.), approx. 480 lbs with packaging
iE33 Specifications
Philips xSTREAM imageformer architecture
Up to 442,176 total digital channels (xMATRIX configuration), 57,000 total digital channels (non xMATRIX configuration)
Displayed Imaging Depth: 1 – 39 cm (transducer dependent)
Up to 180 dB system dynamic range
Philips iE33 Electrical power
Voltage 100V-127V, 220V-240V
Frequency 50/60 Hz
Power consumption: 750VA – 900VA depending on system configuration
Revisions
Philips iE33 revisions: A cart – C cart
Philips first launched the iE33 in 2004 as their new premium shared service ultrasound machine, replacing older units like the Sonos7500. This first version of hardware was designated an “A-cart.” This first version had dual motherboards and dual power supplies and a huge amount of problems with both. The iE33 B-cart improvements were to circuit boards, cables, power supply assembly, and USB ports. The iE33 C-cart brought a new 20” LCD monitor and articulating arm as well as new circuit boards. The R-cart is a A, B, or C-cart with hardware and software upgrades to G4.0 software.
Philips iE33: D cart – F cart
The iE33 D-cart was launched in 2007 and finally fixed the problems with the dual motherboards by using a unified motherboard. D.1 and D.2 included minor hardware updates. The iE33 E-cart made upgrades to the cart, wheels and locking mechanism. E.1 was a minor hardware & software update. E.2 added major power supply improvements to support Matrix transducers. E.3 was a minor update to the way the iE33 was manufactured. The iE33 F-cart was a major upgrade that added Type III channel boards. F.1 was a minor manufacturing update. F.2 upgraded the video card. F.3 upgraded to a new SATA DVD drive.
Philips iE33: G cart
The iE33 G-cart was launched in 2010 and is the latest version of the iE33. This revision added a new channel board and a 21” widescreen monitor. The iE33 G.1 revised the control panel and added larger touch screens. KPI recommends buying D-cart and up as most hardware problems with the iE33 were fixed by this revision and D-carts can be updated to the latest software
Philips iE33 Vision software updates
The software of an iE33 can be updated to a point limited by the hardware cart. Each major revision of the software was designated with the year and “Vision” in the name. “Vision 2012” was the last major release of the software. Very early A-C cart had software that was named simply “system update” with a number. System update 1.0-2.0 have almost all been upgraded over the years and system 3.0 is the lowest version still commonly found on used iE33s.
The Philips iE33 Vision 2007 software update included support for the new L15-7io intraoperative linear transducer, the X7-2 xMatrix pediatric cardiac, and C8-5 microconvex. Tissue specific presets were added to the S5-1 Purewave cardiac sector probe, and the S12-4 and S8-3 were optimized for 2D and Doppler performance. Improvements were made to LVO and QLAB. Features added were TCD (transcranial Doppler) imaging, Fetal Echo Analysis and Live 3D (4D) Echo.
The Philips iE33 Vision 2008 software update included support the new Purewave C5-1 convex transducer. Features added were TMQ (tissue motion quantification) in QLAB, Viewforum stand alone image viewing platform, improved needle visualization, and contrast capabilities.
The Philips iE33 Vision 2009 software update included support for the new S8-3t pediatric micro TEE probe which is 1/3 the size of the S7-2t. Features added were tissue aberration correction, contrast enhanced ultrasound, and SmartExam automation software.
The Vision 2010 software update on the Philips iE33 added xMatrix including support for the new Purewave xMatrix X5-1 transducer, Live xPlane imaging, and 3D Stress Echo, and support for any PACS. (R-carts can have this update, but cannot support the X5-1 transducer.) This iE33 revision also added cardiac diagnostic capabilities related to ischemic disease detection, structural heart disease assessment, as well as systolic and diastolic heart failure and cardiac arrhythmia
The Philips iE33 Vision 2011 software upgraded QLAB with CMQ-Stress replacing TMQ, and with the additions of the Auto Volume tool, GI 3DQ trending, iCrop, and Elevation compounding. Options with Vision 2011 include the xMatrix performance bundle, and Live 3D. (R-carts can have this update.)
The Vision 2012 software on the Philips iE33 adds support for Auto Doppler, Fetal Heart Navigator, and Vascular Plaque Quantification (VPQ).
All revisions of the Philips iE33
Philips iE33 (A.0) with system update 3.0 – Vision 2010 software
Philips iE33 (B.0) with system update 3.0 – Vision 2010 software
Philips iE33 (C.0) with system update 3.0 – Vision 2010 software
Philips iE33 (R.0) with Vision 2007-Vision 2011 software (limited)
Philips iE33 (D.0) with Vision 2007 – Vision 2012 software
Philips iE33 (D.1) with Vision 2007 – Vision 2012 software
Philips iE33 (D.2) with Vision 2007 – Vision 2012 software
Philips iE33 (E.0) with Vision 2007 – Vision 2012 software
Philips iE33 (E.1) with Vision 2007 – Vision 2012 software
Philips iE33 (E.2) with Vision 2007 – Vision 2012 software
Philips iE33 (E.3) with Vision 2007 – Vision 2012 software
Philips iE33 (F.0) with Vision 2007 – Vision 2012 software
Philips iE33 (F.1) with Vision 2007 – Vision 2012 software
Philips iE33 (F.2) with Vision 2007 – Vision 2012 software
Philips iE33 (F.3) with Vision 2007 – Vision 2012 software
Philips iE33 (G.0) with Vision 2011 or Vision 2012 software
Philips iE33 (G.1) with Vision 2011 or Vision 2012 software
Probes
All Philips iE33 probes / transducers
xMATRIX: X3-1 [ 1 – 3 MHz ] 2,400 elements
xMATRIX: X5-1 [ 1 – 5 MHz ] 3,040 elements, PureWave
Pediatric xMATRIX: X7-2 [ 2 – 7 MHz ] 2,500 elements, PureWave
xMATRIX TEE transesophegeal :X 7-2t [ 2 – 7 MHz ] 2,500 elements, PureWave
TEE transesophegeal: S7-2 Omni [ 2 – 7 MHz ] 65 elements, 8.64mm
Pediatric TEE transesophegeal: S7-3t [ 3 – 7 MHz ] 48 elements, 7.63mm,
Pediatric TEE transesophegeal: S8-3t [ 3 – 8 MHz ] 32 elements, 5.2mm shaft diagmeter, multiplane microTEE
ViewFlex Xtra catheter: [ 4.5 – 8.5 MHz ] 64 elements, 120° field of view (for ICE)
ureWave cardiac sector: S5-1 [ 1 – 5 MHz ] 80 elements, 20.3mm, adult cardiac sector
Pediatric cardiac sector S8-3: [ 3 – 8 MHz ] 96 elements, 15.4mm
Neonatal cardiac sector: S12-4 [ 4 – 12 MHz ] 96 elements, 9.78mm
Intraoperative Linear: L15-7io [ 7 – 15 MHz ] 128 elements, 23mm, perioperative
Linear: L11-3 [ 3 – 11 MHz ] 288 elements, 39mm, vascular applications
Linear: L9-3 [ 3 – 9 MHz ] 160 elements, 38mm, vascular applications
Linear:L8-4 [ 4 – 8 MHz ] 128 elements, vascular applications
PureWave Convex: C5-1 [ 1 – 5 MHz ] 160 elements, 96° field of view, Coded beamforming
Microconvex: C8-5 [ 5 – 8 MHz ] 128 elements, 120° field of view, cerebrovascular applications
Microconvex: C9-4 [ 4 – 9 MHz ] 192 elements, 108° field of view, abdominal
Pedoff (CW Transducer): D5cwc [ 5 MHz ] Deep venous and arterial applications, non-imaging
Pedoff (CW Transducer): D2CWC [ 2 MHz ] Adult cardiology applications
Pedoff (PW Transducer): D2tcd [ 2 MHz ] Transcranial Doppler applications, non-imaging
Pulse: TY-306 [ 0.04 – 300 MHz ] Heartsound Pulse Contact Sensor, Cardiac and vascular
Advanced Philips iE33 transducers: Matrix Pediatric
The Philips iE33 not only offers six PureWave single crystal transducers that includes four xMatrix probes. xMatrix not only improves image quality and penetration since xMatrix is developed based on PureWave technology, but also allows for real-time scanning of multiple planes and cardiac 4D without needing a typical mechanical 4D probe. This speeds the acquisition of 4D cardiac data over a 4D mechanical probe for mainly OB/GYN. C-plane resolution is also improved over a typical 4D mechanical probe. The [ 1 – 5 MHz ] X5-1 transducer for the iE33 represents the apex of xMatrix design. The [ 1 – 3 MHz ] X3-1 and [ 2 – 7 MHz ] X7-2 are early xMatrix probes, while the [ 2 – 7 MHz ] X 7-2t is a TEE probe with xMatrix technology and represents the pinnacle of transesephegeal technology. In addition, the iE33 supports the largest number of pediatric cardiac transducers including the [ 3 – 8 MHz ] S8-3t multiplane micro TEE and four other pediatric cardiac transducers. The ViewFlex Xtra catheter developed by St. Jude Medical is also compatible with the iE33 and is used for ICE where the catheter is inserted inside the heart through the left femoral vein.
Popular Philips iE33 transducers
The most popular cardiac sector transducer for the Philips iE33 is the [ 1 – 5 MHz ] S5-1, a PureWave transducer offering better than normal image clarity even at deep penetration. The amazing X 7-2t is the most popular transesphegeal probe for the iE33 with it’s xMatrix 4D and multiplanar capabilities.
Competitors
How the Philips iE33 compares with other Philips systems
TThe Philips iE33 was launched in 2004 as the replacement to the Sonos 7500 and continued in production until 2015. The iE33 is a premium level ultrasound machine that was top of the line till the introduction of the Epiq line in 2013. The Epiq 7 replaced the iE33 and has a larger monitor and larger touchscreen. It also is smaller and lighter than the iE33. The HD15 sits below the iE33 in features, image quality and price and is a shared service ultrasound machine whereas the iE33 is focused only on cardiovascular applications.
Philips iE33 vs iU22
When Philips launched the iE33 it launched the iU22 at the same time and ever since, many medical professionals have wondered what the difference is, and which is best for their needs. The iE33 is focused only on cardio vascular applications where the iU22 is shared service and can be used for women’s health, general imaging as well as cardiac and vascular applications. Because of this the iU22 supports 4D transducers, and several endocaviary transducers, but no neonatal sector or pediatric TEE. The Philips iE33 and iU22 even look nearly identical; with the major distinguishing feature being dual touchscreens for navigation on the iE33 while the iU22 has only one touchscreen.
Other brands competing with the Philips iE33
The Philips iE33 is the premium cardiovascular-focused ultrasound machine of the past decade. Its closest competitor from GE would be the Vivid E9 and both systems are very similar. The iE33 has two touchscreens and a larger monitor compared to the Vivid E9 and also supports ICE in the latest release where the Vivid E9 has no ICE probe or support. The closest competitor from Siemens is the SC2000 which offers similar image quality and features but is stronger in ICE while the iE33 has better xMtrix TEE imaging.
Features
Tissue Harmonic Imaging : Yes
Spatial Compounding(=CrossXbeam) : Yes
Speckle Reduction (=SRI) : Yes
Auto Image Opt(B mode) : Yes
Auto Image Opt(Doppler): Yes
Write Zoom: Yes
Triplex Mode: Yes
Needle Enhancement or Needle Recognition :No
Auto NT Measurement (=Sono NT) :No
Auto Follicle 2D Measurement:No
Auto Follicle 3D Measurement:No
Auto IMT:Yes
Auto IMT (Real Time):No
Automated B/M/D Measurement:Yes
Automated LH Measurement(Automated Function Imaging(AFI), Cardiac Motion Quantification(CMQ), or Auto EF(Ejection Fraction):Yes
Live Dual (B/BC) Mode:Yes
SmartExam or Scan Assistant:Yes
Independent Steer & Lockable Wheels:Yes
Fusion:No
Raw Data File:Yes
Flexible Report:Yes
Barcode Reader:No
Gel Warmer:No
Transducers
Convex (1~6Mhz):Yes(2~5Mhz)
Convex (2~9Mhz):Yes(4~9Mhz)
Single Crystal Convex (1~6Mhz):Yes
Single Crystal Convex (2~9Mhz):No
2D Arrary 3D Convex (1~6Mhz):No
Micro Convex (5~8Mhz):Yes
Single Crystal Endocavity_Straight Type (3~10Mhz):No
Endocavity_Curved Type (5~8Mhz):No
3D Convex (2~6Mhz):No
3D Convex Light Weight (2~7Mhz):No
3D Endocavity (3~10Mhz):Yes
3D Micro Convex (3~9Mhz):No
3D Linear (4~18Mhz):No
Linear (>14Mhz):No
Linear (3~12Mhz):Yes
Linear (<9Mhz):Yes
Single Crystal Linear (>14Mhz):No
Single Crystal Linear (3~12Mhz):No
Single Crystal Linear (<9Mhz):No
Linear 50mm:No
Linear 25mm:No
Hockey stick (<13Mhz):Yes
Hockey stick (>13Mhz):Yes(7~15Mhz io)
T or L shape Intra Operative:Yes
Phased Array_Adult (1~5Mhz):Yes
Single Crystal Phased Array_Adult (1~5Mhz):Yes
2D Arrary 3D Phased Array (1~5Mhz):Yes
Phased Array_Pediatric (3~8hz):Yes
Single Crystal Phased Array_Pediatric (3~8hz):No
Phased Array_Neonate (4~12Mhz):Yes
ICE (Intracardiac Echo Cardiography):No
TEE_Adult (3-7Mhz):Yes
TEE_Pediatric (3~7Mhz):Yes
2D Array 3D TEE (2~7Mhz):Yes
Pencil CW (2Mhz):Yes
Pencil CW (5 or 6Mhz):Yes
Imaging Modes
2D, M mode:Yes
M-color Flow Mode:Yes
Anatomical M-mode:Yes
Trapezoidal Mode:Yes
Color, Power Angio, Pulse Wave Doppler:Yes
Bi-directional Power (=HD FLOW):Yes
SCW Doppler:Yes
Tissue Doppler(Velocity) Imaging:Yes
Freehand 3D:No
Live 3/4D OB/GYN:No
HD Live:No
STIC (Spatio-Temporal Image Correlation):No
Live 3D Echo:Yes
Stress Echo:Yes
Strain and Strain Rate (Cardiac):Yes
B Flow:No
Panoramic Imaging (=Logiq view):Yes
Contrast Imaging – Cardiac:Yes
Contrast Imaging – General Imaging:Yes
Strain-based Elastography:No
Shear Wave Elastography:No
Applications
Abdominal:Yes
Women’s Health Care (GYN & Breast):Yes
OB:Yes
Fetal Echo:Yes
Vascular:Yes
TCD(Transcranial):Yes
Small Parts (Breast, Thyroid, Testis…):Yes
MSK/Anesthesiology:Yes
Pediatrics:Yes
Urology (Renal, Prostate…):Yes
Echocardiography_Adult:Yes
Interventional Cardiology:Yes
Echocardiography_Pediatric:Yes
Echocardiography_Neonate:Yes
Stress Echocardiography:Yes
Transesophageal Echo_Adult:Yes
Transesophageal Echo_Pediatric:No
Internal Medicine w/ Shared Service:Yes
Surgury:Yes
Interventional Radiology:Yes
Contrast Imaging _ General Imaging (Low MI):Yes
Contrast Imaging _ Cardiac (High or Low MI): Yes
Bowel Imaging:Yes
Strain Elastography:Yes
Shear Wave Elastography:No
Specification
System Overview
Year Launched: 2004
Estimated Market Price ($): Premium
Monitor (inch): 17″LCD(20″ option)
Tilt/Rotate Adjustable Monitor: Yes
Monitor Resolution:
Image Size Resolution:
Touch Screen (Inch): DUAL
Trackball or Trackpad: Trackball
CP Back-Lighting:Yes
Weight:345lbs
Probe Ports: 3
Battery:No
Boot-Up Time:120sec
Sleep Mode (Quick Start):30sec(stanby mode)
Maximum Depth of Field:30cm
Minimum Depth of Field:2cm
Cart (HCU):No
Independent Steer & Lockable Wheels:Yes
Connectivity
DICOM 3.0:Yes(Option)
DICOM SR_Cardiac:Yes
DICOM SR_Vascular :Yes
DICOM SR_OB/GYN:Yes
JPEG, WMV, & AVI:Yes
USB:Yes(4)
HDD/SDD:160GB
DVD/CD RW:Yes
Wireless LAN:No
Shipping Policy
Orders made at Medpick are initiated and processed for shipment upon receipt of request from the customer. Please note that our Shipping Services (Fee, Transportation, Loss or Damage of any shipment, etc.) are in accordance with the Seller\'s terms of Shipment.
Refund Policy
Please refer to Medpick Return Policy.
Cancellation / Return / Exchange Policy
Please refer to Medpick Return Policy.
 REGISTER
REGISTER
 SIGN IN
SIGN IN